Thailand to Resume AstraZeneca Jabs Tomorrow
Share:
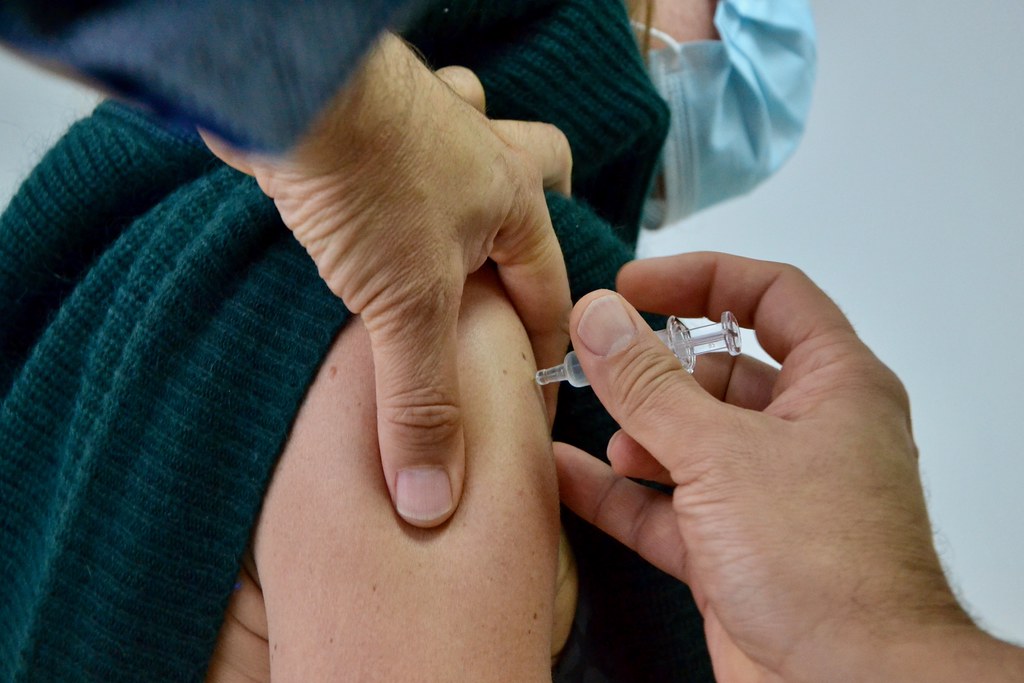
The Kingdom will resume vaccinations using the AstraZeneca vaccine after its usage was suspended last Friday over reports of blood clots developed by recipients in Denmark. According to health officials, Prime Minister Prayut Chan-o-cha and his Cabinet members who are over 60 years of age will be the first recipients.
Authorities in Denmark had declared a suspension on the use of the AstraZeneca vaccine in its immunization program after a number of cases, including one fatality, wherein the patient developed blood clots after receiving the vaccine.
Norway, Iceland and the Netherlands followed suit. In Asia, Thailand was the first country to declare a halt on the use of the vaccine pending an investigation by the European Medicines Agency or EMA.
Health authorities in Thailand said that they are halting the use of AstraZeneca’s vaccine pending an update on the cases.
Today, however, government house spokesperson Natreeya Thaweewong said that they will push through with the delayed vaccination of the Prime Minister and the Cabinet tomorrow, March 16.
This followed statements earlier today by Public Health Minister Anutin Charnvirakul that, once local health experts give the clearance for the vaccine, they can resume use of the vaccine in the immunization program.
According to Minister Anutin, several countries had confirmed that they experienced no issues with blood clots and the vaccine, and that they will continue administering it despite the temporary halt in European countries.
The Minister said that the academic committee on vaccines will be meeting this afternoon to discuss the issue, and give the go signal if there were no further information from abroad.
The World Health Organization had insisted on Friday that the AstraZeneca vaccine was safe and an ideal choice for vaccinations. The WHO added that there was no direct causal link between the blood clots and the vaccine.
The manufacturer of the vaccine itself said that a “careful review” of data from clinical trials had showed there was no evidence in their studies that recipients are at risk of developing pulmonary embolism, deep vein thrombosis and thrombocytopenia.
Medical experts in Thailand also said that the delay in the use of the vaccine is not indicative the vaccine was problematic, but rather was intended to give way to investigations to verify if there is indeed a link between the shots and the blood clots that were reported.
AstraZeneca will be providing the bulk of the vaccines that the Kingdom will be using for its immunization program for the general population. Under a transfer of technology agreement, Siam Biotech will be locally producing the vaccine starting in June.
Siam Biotech will be rolling out a total of 61 million doses for the second phase of the immunization program.
The first phase will use more than 100,000 doses of the AstraZeneca and 2 million doses of the Chinese vaccine maker Sinovac’s jabs.
An initial batch of 200,000 doses had arrived this month and were already distributed to high priority recipients. Another batch of 800,000 doses is due to arrive on March 20, with the final 800,000 doses arriving next month.
Source:
BangkokPost